Characterization of CD8+T cell responses during H. pylori infection
Strong Th1 and Th17 responses have been observed in the stomach of H. pylori infected individuals. However, the bacterium has developed different strategies in order to at the same time elicit regulatory T cell responses that contribute to immune escape. Although studies in human and mice have focused on CD4+-T cell-mediated responses, data from different human clinical studies have indicated a potential relevance of CD8+ cytotoxic lymphocytes in the immune response to H. pylori infection. An association between increased CD8+T cells in response to H. pylori colonization and development of gastric ulcer has been observed in infected patients. In mice, we have shown that H. pylori induces strong recruitment of CD8+T cells into the stomach, which highly contribute to the production of IFNγ in response to the infection. However, the nature and the role of CD8+T cells during H. pylori infection and bacterial determinants recognized by these cells remain unclear. Therefore, we are conducting an exhaustive characterization of T cell responses over time upon H. pylori infection in murine stomach as well as in human gastric biopsies. By using peptide libraries from different virulence factors, we aim at identifying peptides eliciting anti-H. pylori specific CD8 T cell responses.
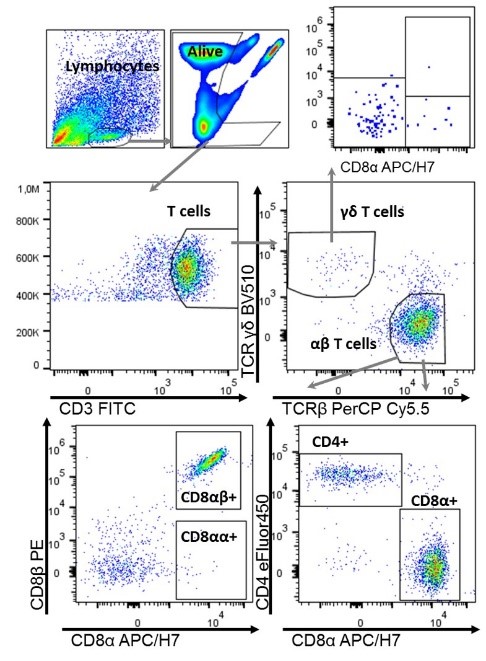
Characterization of CD8+ T cells upon H. pylori infection by flow cytometry.
Publications
Koch MRA, Gong R, Friedrich V, Engelsberger V, Kretschmer L, Wanisch A, Jarosch S, Ralser A, Lugen B, Quante M, Vieth M, Vasapolli R, Schulz C, Buchholz VR, Busch DH, Mejías-Luque R, Gerhard M: CagA-specific gastric CD8+ tissue-resident T cells control Helicobacter pylori during the early infection phase. Gastroenterology (2023); doi: https://doi.org/10.1053/j.gastro.2022.12.016(link is external), (Full text(link is external), pubmed(link is external))
Wüstner S, Anderl F, Wanisch A, Sachs C. Steiger K, Nerlich A, Vieth M, Mejías-Luque R, Gerhard M. Helicobacter pylori γ-glutamyl transferase contributes to colonization and differential recruitment of T cells during persistence. Scientific Reports volume 7, Article number: 13636 (2017). (Full Text; Abstract)


